Notice: Morris Bart is no longer accepting new cases related to Zostavax.
From 2006 to 2017, Zostavax was the only shingles vaccine available in the US. But after a decade in use, over 100,000 patients have reported problems with the vaccine to the FDA. Here’s the important information you need to know. Unfortunately, in December 2022, the MDL judge dismissed these cases. This decision is currently being appealed, and the MDL will stay open pending the resolution of that appeal.
What Is Zostavax?
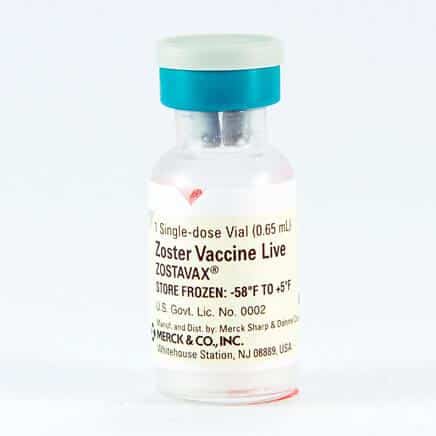
The Zostavax vaccine introduces a weakened version of the shingles virus to the body, encouraging it to develop antibodies against the virus and, therefore, strengthen the immune system. Zostavax is a live attenuated virus vaccine used to prevent shingles (also known as herpes zoster) in older adults. The “shingles vac” or “zoster vaccination” is manufactured by Merck and was first approved by the FDA for use in 2006. Until 2017 Zostavax was the FDA preferred shingles vaccine.
Zostavax is administered as a one-time shot in a doctor’s office or pharmacy. Although the FDA claimed that the Zostavax vaccination was 51% effective at preventing shingles, in 2017, a new vaccine was introduced that is now the FDA’s preferred method of preventing shingles.
FACT CHECK
What Causes Shingles?
Shingles occur when the varicella zoster virus (the same virus that causes chickenpox) is reactivated. Therefore, people with a weakened immune system are the most likely to develop shingles.
FACT CHECK
What Are Shingles Symptoms?
Shingles is a painful skin rash often accompanied by redness, pain, numbness, burning, and sometimes blisters.
Who Has Been Exposed to Zostavax?
Until a new vaccine was introduced in 2017, the FDA recommended that all adults over 60 receive the vaccine. However, many patients with weakened immune systems received the vaccine once they turned 50 as a precautionary measure. Although it is impossible to know how many older Americans were exposed to Zostavax, there is a good chance that if you received a shingles vaccine shot in the past decade, you may have been exposed to Zostavax.
What Are the Side Effects of the Shingles Vaccine?
The most commonly reported side effects of Zostavax are headache and injection site reactions, including swelling, pain, redness, and bruising. However, since the vaccine was introduced in 2006, the FDA has received over 100,000 reports of unexpected or serious side effects. These side effects can take minutes to months to develop, and some even resemble the symptoms of the shingles that the vaccine was intended to prevent.
After receiving Zostavax, the most commonly reported side effects include:
- Joint pain
- Fever
- Muscle pain
- Chickenpox
- Nausea
- Swollen glands near the vaccine injection site
- Shingles
Alarmingly, some patients reported even more serious side effects after receiving the shingles vaccine, including:
- Allergic reactions
- Hearing loss
- Severe eye infections and/or vision loss (Acute retinal necrosis)
- Autoimmune disorders
- Nerve pain or damage (Post-herpetic neuralgia)
- Congestive heart failure
- Spinal cord inflammation
- Death
What You Need to Know about Shingles Vaccine Lawsuits
As more older Americans begin to experience serious Zostavax side effects, some patients choose to report these problems to the FDA and then file suit against the vaccine’s manufacturer, Merck. Although many of these lawsuits have just begun to work their way through the court system, in August 2018, a Pennsylvania federal court consolidated several of these claims into one case, MDL 2848, to promote efficiency and fairness in the face of mounting claims against Merck.
Do I have a Zostavax Case?
Unfortunately for those who may have been eligible for a Zostavax lawsuit, the MDL judge dismissed these cases in December 2022. While this decision is currently being appealed, it is a bad sign for all those impacted by Zostavax. The MDL will stay open pending the resolution of that appeal.